What Is Peptide Therap? Ay Medical Provider’s Guide to Benefits, Risks, and Results
Evidence-based insights on peptide therapy mechanisms, clinical applications, and safety protocols for medical providers.
Core Mechanisms
Peptides are amino acid chains that bind to specific receptors, triggering targeted biological responses like tissue repair, metabolic regulation, and immune modulation without broad systemic effects.Clinical Benefits
Proven applications include weight management, wound healing, immune optimization, cognitive support, and recovery enhancement with measurable outcomes across multiple medical specialties.Safety Protocols
Requires thorough patient screening, baseline lab work, regular monitoring, and informed consent addressing off-label use and evidence quality for each peptide compound.
Peptide therapy represents a rapidly evolving field where clinical promise meets regulatory complexity. As a healthcare provider, you need clarity on what works, what's safe, and how to implement protocols that deliver measurable outcomes. This guide provides the evidence-based framework you need to integrate peptide therapy into your practice with confidence and clinical precision.
Understanding Peptide Therapy: Core Mechanisms and Medical Applications
Patient Evaluation Protocol
Laboratory Testing
Complete blood count and metabolic panel
Physical Examination
Document vitals and body composition
Medical History
Review contraindications and interactions
Protocol Selection
Choose appropriate peptide therapy
Peptide therapy involves administering specific amino acid sequences to trigger targeted biological responses in the body.
These short-chain amino acids—typically containing 2-50 amino acid units—act as signaling molecules that communicate with cells to regulate physiological processes.
Unlike traditional pharmaceuticals that often block or inhibit pathways, peptides work by mimicking natural biological signals your body already recognizes.
The therapeutic mechanism operates through receptor binding. When a peptide encounters its target receptor on a cell surface, it initiates a cascade of intracellular events.
This specificity allows for precise modulation of functions like hormone production, immune response, tissue repair, and metabolic regulation.
In the United States, peptide therapy represents a growing area of clinical interest within preventive and regenerative medicine, with applications explored in structured programs related to metabolic support, age-related physiological changes, hormonal balance, musculoskeletal recovery, and general vitality optimization.
Medical providers increasingly utilize peptides because they offer high selectivity with typically fewer off-target effects compared to small-molecule drugs.
Classification of Therapeutic Peptides
Therapeutic peptides fall into several functional categories. Growth factor peptides like BPC-157 and TB-500 promote tissue regeneration and wound healing. Metabolic peptides such as GLP-1 agonists regulate insulin secretion and appetite control. Immune-modulating peptides including thymosin alpha-1 enhance immune system function. Neuropeptides like cerebrolysin support cognitive function and neurological health.
Delivery Methods and Bioavailability Considerations
Peptide administration routes significantly impact therapeutic outcomes.
Subcutaneous injection remains the most common delivery method, delivering peptides just under the skin and allowing slow absorption and consistent therapeutic effects while minimizing discomfort.
Intramuscular injection offers slower release kinetics for certain peptides. Oral administration faces challenges due to gastric degradation, though some peptides utilize protective formulations or are specifically designed for oral stability.
Transdermal and nasal delivery routes are emerging for specific peptides, offering non-invasive alternatives. Bioavailability varies dramatically by peptide structure and delivery method—subcutaneous administration typically achieves 70-90% bioavailability, while oral routes may drop below 10% for unprotected peptides.
Regulatory Status and Clinical Validation
The regulatory landscape for peptide therapy varies by compound.
On February 27, 2026, HHS Secretary Robert F. Kennedy Jr. announced that approximately 14 of the 19 peptides previously placed on the FDA’s Category 2 “do not compound” list would be moved back to Category 1 status, meaning licensed compounding pharmacies across the United States could soon legally prepare these peptides again for patients with valid prescriptions.
Some peptides hold FDA approval for specific indications—semaglutide for diabetes and weight management, and sermorelin was FDA-approved in 1997 for growth hormone deficiency in children, though the manufacturer discontinued production in 2008 for commercial reasons.
Many peptides used in clinical practice operate under research or compounding pharmacy frameworks, requiring informed consent and careful documentation.
Clinical validation levels differ substantially across peptides. Some possess extensive randomized controlled trial data, while others rely on preliminary studies, animal models, or observational evidence. Medical providers must critically evaluate the evidence base for each peptide, distinguishing between well-established therapies and emerging applications with limited human data.
Benefits and Clinical Outcomes Across Medical Specialties
Peptide therapy delivers measurable clinical benefits across multiple medical domains. The therapeutic versatility stems from peptides’ ability to target specific physiological pathways without the broad systemic effects typical of many conventional medications. Healthcare providers at facilities like Nebraska Wellness Group integrate peptide protocols into comprehensive treatment plans for patients seeking optimized health outcomes.
Metabolic Health and Weight Management
GLP-1 receptor agonists and related peptides demonstrate significant efficacy in metabolic regulation. These compounds enhance insulin sensitivity, reduce glucagon secretion, and slow gastric emptying. Clinical outcomes include sustained weight reduction, improved glycemic control in diabetic patients, and favorable changes in cardiovascular risk markers.
Patients typically experience appetite suppression within the first week of treatment, with progressive weight loss over 12-24 weeks.
Average weight reduction ranges from 10-15% of initial body weight when combined with lifestyle modifications, with oral semaglutide demonstrating a mean change of -13.6% in body weight from baseline to week 64 compared with -2.2% in the placebo group.
Metabolic improvements often include reduced hemoglobin A1c levels, decreased triglycerides, and improved blood pressure readings.
Tissue Repair and Wound Healing
Regenerative peptides accelerate healing processes through multiple mechanisms.
BPC-157 activates several overlapping pathways, notably VEGFR2 and nitric oxide synthesis via the Akt-eNOS axis, promoting angiogenesis, fibroblast activity, and neuromuscular stabilization, particularly in poorly vascularized tissues such as tendons and myotendinous junctions.
TB-500 facilitates cell migration and reduces inflammation. These properties make them valuable for chronic wounds, post-surgical recovery, and musculoskeletal injuries.
Healthcare providers utilize these peptides for diabetic ulcers, surgical incisions, tendon injuries, and ligament damage. The anti-inflammatory properties complement the regenerative effects, creating an optimal healing environment. Treatment duration typically spans 4-12 weeks depending on injury severity.
Immune System Optimization
Immune-modulating peptides like thymosin alpha-1 can stimulate the proliferation, differentiation, and maturation of T cells in the thymus, which may elevate CD4+ T lymphocyte levels and bolster the CD4+/CD8+ ratio, thereby enhancing immune function.
Medical applications include chronic viral infections, immunodeficiency states, and as adjunctive therapy during cancer treatment. These peptides don’t stimulate the immune system indiscriminately but rather restore balanced immune function.
Patients with recurrent infections often show reduced infection frequency and severity. Those with autoimmune conditions may experience modulation of overactive immune responses. Treatment protocols typically involve cycles of 4-8 weeks with monitoring of immune markers.
Cognitive Function and Neuroprotection
Neuropeptides support brain health through neurotrophic effects, improved cerebral blood flow, and neuroprotection against oxidative stress. Applications include cognitive decline, traumatic brain injury recovery, and neurodegenerative disease management. Cerebrolysin and similar compounds demonstrate benefits in memory, processing speed, and executive function.
Clinical outcomes vary based on baseline cognitive status. Patients with mild cognitive impairment show more dramatic improvements than those with advanced neurodegeneration. Treatment courses often extend 3-6 months for optimal neuroplastic changes.
Performance Enhancement and Recovery
Athletic and active populations utilize peptides for recovery optimization and performance support. Growth hormone secretagogues like sermorelin and ipamorelin enhance natural growth hormone production, supporting muscle recovery, sleep quality, and body composition. These peptides offer an alternative to direct growth hormone administration with a more physiological effect profile.
Recovery times between training sessions typically decrease, while muscle soreness and injury rates may improve. Body composition changes include modest increases in lean mass and reductions in body fat percentage over 3-6 month protocols.
Risk Assessment and Contraindication Management
Absolute Contraindications
Growth-promoting peptides may accelerate tumor growth. Insufficient safety data exists for pregnant/nursing women.
Understanding the risk profile of peptide therapy enables informed clinical decision-making and appropriate patient selection. While peptides generally demonstrate favorable safety profiles compared to many pharmaceutical agents, specific risks and contraindications require careful consideration. Thorough patient screening and ongoing monitoring form the foundation of safe peptide therapy implementation.

Common Adverse Effects and Management Strategies
Most peptide-related adverse effects are mild and transient.
Injection site reactions—including redness, swelling, or discomfort—occur in approximately 20-40% of subjects with subcutaneous administration in Phase I trials.
These typically resolve within 24-48 hours and can be minimized through proper injection technique, site rotation, and temperature management of peptide solutions.
Gastrointestinal symptoms affect certain peptide classes more than others. GLP-1 agonists commonly cause nausea, reduced appetite, and occasional vomiting, particularly during dose titration. Starting at lower doses and advancing gradually reduces these effects significantly. Most patients develop tolerance within 2-4 weeks.
Hormonal peptides may cause temporary hormonal fluctuations. Growth hormone secretagogues occasionally produce mild fluid retention, joint discomfort, or transient changes in glucose metabolism. These effects typically normalize as the body adjusts to treatment or with dose modification.
Serious Risks and Clinical Monitoring Requirements
Though uncommon, serious adverse events warrant vigilant monitoring. Hypersensitivity reactions range from mild allergic responses to rare anaphylaxis. Patients with multiple drug allergies or history of anaphylaxis require heightened caution and may benefit from test dosing protocols.
Metabolic disturbances can occur with certain peptides. Growth hormone-related compounds may affect glucose homeostasis, requiring regular blood glucose monitoring in diabetic or pre-diabetic patients. Thyroid function occasionally shifts with long-term peptide use, necessitating periodic thyroid panel assessment.
| Risk Category | Monitoring Frequency | Key Parameters |
|---|---|---|
| Metabolic | Every 3 months | Glucose, HbA1c, lipid panel |
| Hormonal | Every 3-6 months | IGF-1, thyroid panel, sex hormones |
| Renal | Every 6 months | Creatinine, eGFR, urinalysis |
| Hepatic | Every 6 months | AST, ALT, bilirubin |
| Cardiovascular | Baseline + as indicated | Blood pressure, ECG if indicated |
Absolute and Relative Contraindications
Absolute contraindications include active malignancy for growth-promoting peptides, as these compounds may theoretically accelerate tumor growth. Pregnant or breastfeeding women should avoid peptide therapy due to insufficient safety data. Patients with known hypersensitivity to specific peptides or their excipients must avoid those formulations.
Relative contraindications require individualized risk-benefit assessment. Advanced kidney disease affects peptide clearance and may necessitate dose adjustments or alternative therapies. Uncontrolled diabetes presents challenges for certain metabolic peptides. Severe cardiovascular disease requires careful evaluation before initiating peptides affecting fluid balance or cardiac workload.
Drug Interactions and Combination Therapy Considerations
Peptides interact with conventional medications through various mechanisms. GLP-1 agonists slow gastric emptying, potentially affecting absorption of oral medications requiring rapid onset. Timing adjustments or alternative formulations may be necessary for critical medications.
Growth hormone secretagogues may alter insulin sensitivity, requiring adjustment of diabetic medications. Immunomodulatory peptides could theoretically interact with immunosuppressants, though clinical significance remains unclear. Always review complete medication lists before initiating peptide therapy.
Patient Selection Criteria and Informed Consent
Appropriate patient selection minimizes risk and optimizes outcomes. Ideal candidates demonstrate realistic expectations, commitment to monitoring protocols, and stable baseline health status. Patients should understand that peptide therapy often represents an adjunctive approach rather than standalone treatment.
Comprehensive informed consent must address off-label use status for non-FDA-approved indications, evidence quality, potential risks, monitoring requirements, and cost considerations.
Documentation should reflect detailed discussions about alternative treatments, expected outcomes, and the evolving nature of peptide therapy research. Patients must commit to regular follow-up appointments and laboratory monitoring as clinically indicated.
Evidence-Based Treatment Protocols and Dosing Strategies
Implementing peptide therapy requires structured protocols based on available clinical evidence and pharmacokinetic principles. Dosing strategies balance therapeutic efficacy against tolerability, with individualization based on patient characteristics, treatment goals, and response patterns. Medical providers must understand both standard protocols and appropriate modifications for specific clinical scenarios.
Initial Assessment and Baseline Evaluation
Comprehensive patient evaluation prior to peptide therapy initiation constitutes the foundation of clinical safety protocols, with laboratory baseline measurements including complete blood count, comprehensive metabolic panel, thyroid function tests, and hormone profiles relevant to the specific peptide therapeutic being considered.
For growth hormone secretagogues, baseline IGF-1 levels and glucose tolerance assessment are mandatory.
Physical examination documents baseline vital signs, body composition measurements, and condition-specific assessments. For wound healing applications, photograph and measure wounds. For cognitive applications, establish baseline cognitive testing. For metabolic therapy, record weight, body mass index, and waist circumference.
Medical history review identifies contraindications, potential drug interactions, and factors affecting treatment selection. Document previous medication responses, allergies, surgical history, and family medical history. This foundation enables informed protocol selection and appropriate safety monitoring.
Peptide-Specific Dosing Frameworks
GLP-1 Receptor Agonists follow titration schedules to minimize gastrointestinal side effects. Semaglutide typically starts at 0.25 mg weekly, advancing to 0.5 mg after four weeks, then potentially to 1.0 mg or higher based on tolerance and response. Dose escalation occurs no more frequently than monthly, allowing physiological adaptation.
Growth Hormone Secretagogues like sermorelin typically begin at 200-300 mcg daily via subcutaneous injection, administered before bedtime to align with natural growth hormone pulsatility. Ipamorelin doses range from 200-300 mcg once or twice daily. Treatment cycles often span 3-6 months with periodic reassessment.
Regenerative Peptides such as BPC-157 commonly utilize 250-500 mcg twice daily, administered subcutaneously near the injury site or systemically. TB-500 loading phases may employ 2-2.5 mg twice weekly for 4-6 weeks, followed by maintenance dosing of 2-2.5 mg weekly or biweekly.
Administration Timing and Frequency Optimization
Timing significantly impacts peptide efficacy. Growth hormone secretagogues demonstrate optimal results with evening administration, capitalizing on natural nocturnal growth hormone pulses. Metabolic peptides like GLP-1 agonists may be dosed weekly, while others require daily administration.
Regenerative peptides benefit from consistent dosing intervals—typically twice daily for optimal tissue concentrations. Some protocols employ higher frequency during acute phases, transitioning to maintenance schedules as healing progresses. Consistency matters more than perfection; missing occasional doses is less problematic than erratic administration patterns.
Response Monitoring and Protocol Adjustment
Clinical response assessment combines subjective patient reports with objective measurements. Weight management protocols track weekly weight, monthly body composition analysis, and quarterly metabolic panels. Regenerative protocols document healing progression through photography, measurement, and functional assessments.
Laboratory monitoring intervals depend on peptide class and patient risk factors, with a recommended schedule to test hormone levels and key markers at 1 month, 3–6 months, and annually thereafter.
Growth hormone therapy requires IGF-1 monitoring every 3 months during titration, then every 6 months during maintenance. Metabolic peptides necessitate glucose and lipid monitoring every 3 months initially, with interval extension once stable.
Dose adjustments should occur methodically based on clear criteria: inadequate response after adequate trial duration, intolerable side effects, or laboratory abnormalities requiring intervention.
Combination Therapy Approaches
Strategic peptide combinations may enhance outcomes beyond monotherapy. Growth hormone secretagogues often pair with metabolic peptides for comprehensive body composition optimization. Regenerative peptides combine effectively with immune-modulating compounds for complex wound healing scenarios.
Combination protocols require careful consideration of overlapping effects and cumulative risks. Starting peptides sequentially rather than simultaneously enables clearer attribution of effects and side effects. Monitor more frequently when combining peptides with conventional medications affecting similar pathways.
Treatment Duration and Discontinuation Strategies
Optimal treatment duration varies by indication and individual response. Metabolic peptides for weight management typically continue until goal achievement, followed by maintenance phases or gradual discontinuation with lifestyle support. Regenerative protocols conclude when healing objectives are met, usually 8-16 weeks.
Growth hormone optimization may involve longer-term therapy with periodic breaks to assess endogenous function recovery. Discontinuation should be gradual when appropriate, particularly for peptides affecting hormonal axes. Abrupt cessation rarely causes withdrawal symptoms but may result in gradual return to baseline function over weeks to months.
Integration into Clinical Practice: Implementation Framework
How Peptides Work
Short-chain amino acids act as signaling molecules that bind to cell receptors, triggering targeted biological responses by mimicking natural signals your body already recognizes.
Core mechanism explanationPeptide Categories
4 categories- 1 Growth factor peptides: tissue regeneration
- 2 Metabolic peptides: insulin and appetite control
- 3 Immune-modulating peptides: enhance immunity
- 4 Neuropeptides: support cognitive function
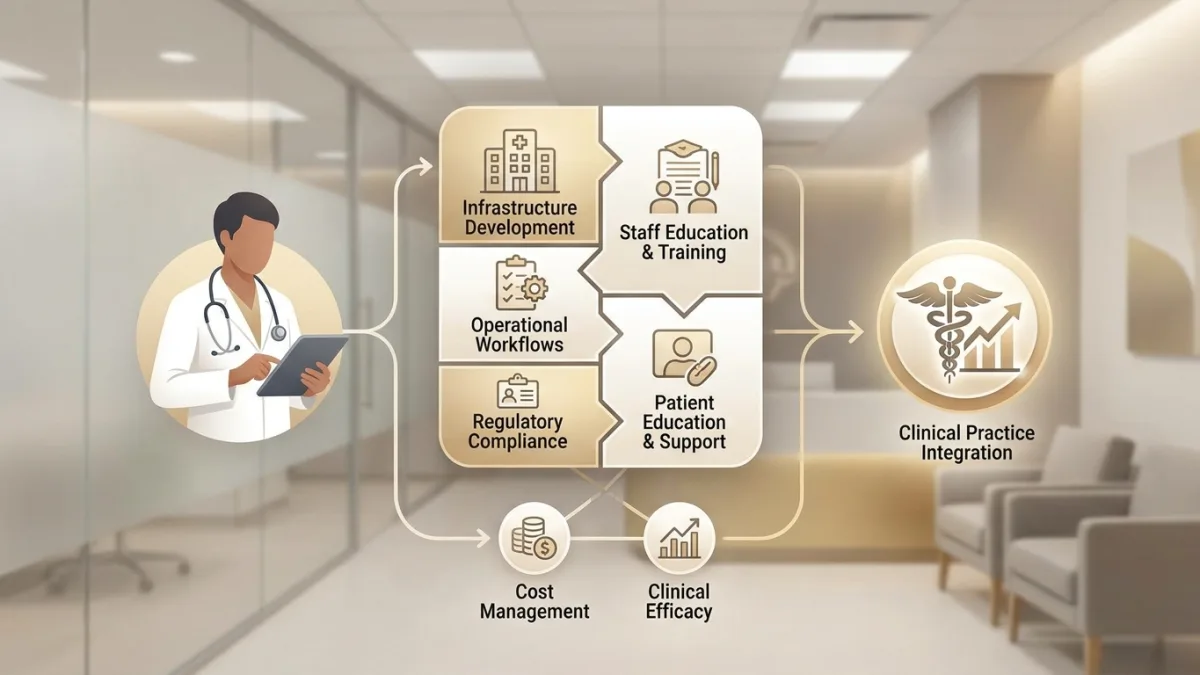
Successfully incorporating peptide therapy into medical practice requires systematic infrastructure development, staff education, and operational workflows. Healthcare providers must balance clinical efficacy with practical considerations including regulatory compliance, cost management, and patient education. Facilities like Nebraska Wellness Group demonstrate how peptide therapy integrates into comprehensive patient care models.
Practice Setup and Regulatory Compliance
Establishing peptide therapy services begins with regulatory framework understanding.
According to the Federal Food, Drug, and Cosmetic Act (FDCA), any substance intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease must be approved by the FDA before it can be marketed, with the FDA categorizing peptides as biologics requiring them to meet specific safety and efficacy standards before approval.
Peptides prescribed for FDA-approved indications follow standard prescribing protocols. Off-label applications require enhanced informed consent documentation and medical justification in patient records.
Compounded peptides necessitate relationships with licensed compounding pharmacies that comply with FDA and United States Pharmacopeia (USP) standards, with guidelines referencing USP Chapter 797 which outlines sterile compounding practices.
State medical board regulations vary regarding peptide prescribing. Some jurisdictions require specific training or certification for certain peptide classes. Maintaining current knowledge of regulatory changes protects both providers and patients. Consider consulting healthcare attorneys familiar with regenerative medicine regulations during practice setup.
Documentation standards should exceed minimum requirements. Record detailed clinical rationale, alternative treatments considered, informed consent discussions, baseline assessments, treatment protocols, monitoring plans, and outcome tracking. Comprehensive documentation supports medical necessity and protects against regulatory scrutiny.
Staff Training and Competency Development
Clinical staff require specialized training in peptide reconstitution, storage, handling, and administration techniques.
Proper injection technique minimizes adverse effects and optimizes absorption, with improper administration being one of the most common causes of preventable complications, making it essential for providers to teach safe, clear instructions since “how to inject peptides” is a top patient search term.
Staff should demonstrate competency in subcutaneous and intramuscular injection, site selection, and sterile technique before patient contact.
Knowledge training encompasses peptide mechanisms, expected effects, common side effects, and emergency response protocols. Staff should confidently answer common patient questions and recognize situations requiring provider consultation. Regular continuing education maintains current knowledge as peptide research evolves.
Patient Education Materials and Communication Strategies
Comprehensive patient education materials enhance compliance and satisfaction. Written protocols should detail reconstitution instructions with photographs, injection technique guides, storage requirements, and troubleshooting common issues. Video demonstrations supplement written materials effectively.
Educational content should address realistic expectations, typical timelines for results, potential side effects and management, monitoring requirements, and cost considerations. Patients better tolerate temporary side effects when they understand these represent normal responses rather than treatment failure.
Supply Chain Management and Quality Assurance
Peptide quality varies substantially across suppliers. Source peptides from reputable compounding pharmacies or pharmaceutical manufacturers with third-party testing verification. Certificate of analysis documentation confirms peptide purity, concentration, and sterility. Avoid gray-market suppliers lacking quality assurance.
Storage requirements demand attention—most peptides require refrigeration, while some need freezing. Reconstituted peptides have limited stability, typically 30-90 days refrigerated depending on formulation. Implement inventory management systems preventing expired product dispensing. Temperature monitoring of storage areas protects peptide integrity.
Financial Considerations and Patient Access
Peptide therapy costs create access barriers for some patients. Insurance coverage remains limited, with most peptides requiring out-of-pocket payment. Transparent pricing discussions during initial consultations prevent misunderstandings. Consider tiered pricing structures or payment plans to improve accessibility.
Cost-effectiveness analysis helps patients make informed decisions. Compare peptide therapy costs against alternative treatments, considering both direct expenses and indirect costs like time off work or additional medications. Some peptides offer superior cost-effectiveness despite higher upfront costs through reduced need for other interventions.
Frequently Asked Questions
Peptide therapy involves using short chains of amino acids that act as cell-signaling molecules to regulate important biological functions. These peptides bind to specific receptors in your body and trigger targeted responses such as hormone production, tissue repair, and metabolic enhancement. Unlike traditional medications that often suppress symptoms, peptides work by optimizing your body’s natural processes over time. Medical supervision ensures safe administration and proper dosing for your individual health goals.
Most patients notice initial effects within 2 to 6 weeks of starting treatment. However, significant long-term changes such as fat loss, enhanced metabolism, and improved muscle tone typically take several weeks to a few months to become noticeable, with significant improvements appearing over 3 to 6 months. The timeline varies based on the specific peptide used, your individual biology, treatment consistency, and overall health status. Patience and adherence to your prescribed treatment plan are essential for optimal outcomes.
Common side effects include injection site reactions, mild fatigue, water retention, and digestive changes. More serious but rare risks include hormonal imbalances and allergic reactions. Working with a qualified healthcare provider who monitors your progress minimizes these risks significantly.
Costs vary based on the specific peptide type, dosage, treatment frequency, and provider expertise. Most insurance plans do not cover peptide therapy for wellness purposes, though some may provide partial coverage if deemed medically necessary.
Peptides act on hormone pathways and require proper medical evaluation to prevent serious complications. Medical supervision is critical because peptides can influence multiple physiological systems simultaneously, and improper dosing or sourcing can lead to hormonal imbalances, adverse drug interactions, and contamination risks. A licensed physician reviews your medical history, conducts necessary lab testing, monitors your response, and adjusts treatment protocols to ensure safety and effectiveness. This oversight protects you from the dangers of unregulated online sources and self-administration.
Individuals with active cancer, kidney or liver disease, uncontrolled diabetes, cardiovascular disorders, autoimmune conditions, or those who are pregnant or breastfeeding should avoid peptide therapy or use it only under strict medical supervision. Always consult a qualified healthcare provider before starting any peptide protocol.
In February 2026, approximately 14 of the 19 peptides previously placed on the FDA’s Category 2 restricted list were announced to be moved back to Category 1 status, allowing licensed compounding pharmacies to legally prepare them when prescribed by a physician. This reclassification allows peptides like BPC-157, Thymosin Alpha-1, AOD-9604, CJC-1295, and Ipamorelin to be compounded legally under medical supervision. However, Category 1 status is not the same as FDA approval—these remain prescription therapeutics used under physician discretion. Some peptides have full FDA approval for specific medical conditions, while others are available through regulated compounding pharmacies.

